Current Clinical Trials
Now Enrolling: Oncology Clinical Trials at Salib Oncology
We are currently enrolling participants for IRB-approved clinical research studies.
Qualified participants may receive study-related care and compensation. Participation is voluntary, and all inquiries are handled confidentially.
Please review the studies below to see if you may qualify.
Limited-Stage Small-Cell Lung Cancer (LS-SCLC)
Phase 3 Study of Tarlatamab Therapy
Who May Qualify:
- Diagnosed with limited-stage small-cell lung cancer
- Completed chemotherapy and radiation
- No disease progression
Purpose: Evaluating treatment after standard therapy.
Extensive-Stage Small-Cell Lung Cancer (ES-SCLC)
Phase 3 Study of Sacituzumab Govitecan
Who May Qualify:
- Diagnosed with extensive-stage small-cell lung cancer
- Prior platinum-based chemotherapy
- Disease progression
Purpose: Comparing investigational therapy with standard care.
Advanced or Metastatic Non-Squamous NSCLC
Phase 2 Study of ABBV-400 + Budigalimab
Who May Qualify:
- Diagnosed with advanced or metastatic non-small cell lung cancer
- No prior treatment for advanced disease
- No actionable mutations
Purpose:
Evaluating combination therapy as a first-line treatment option.
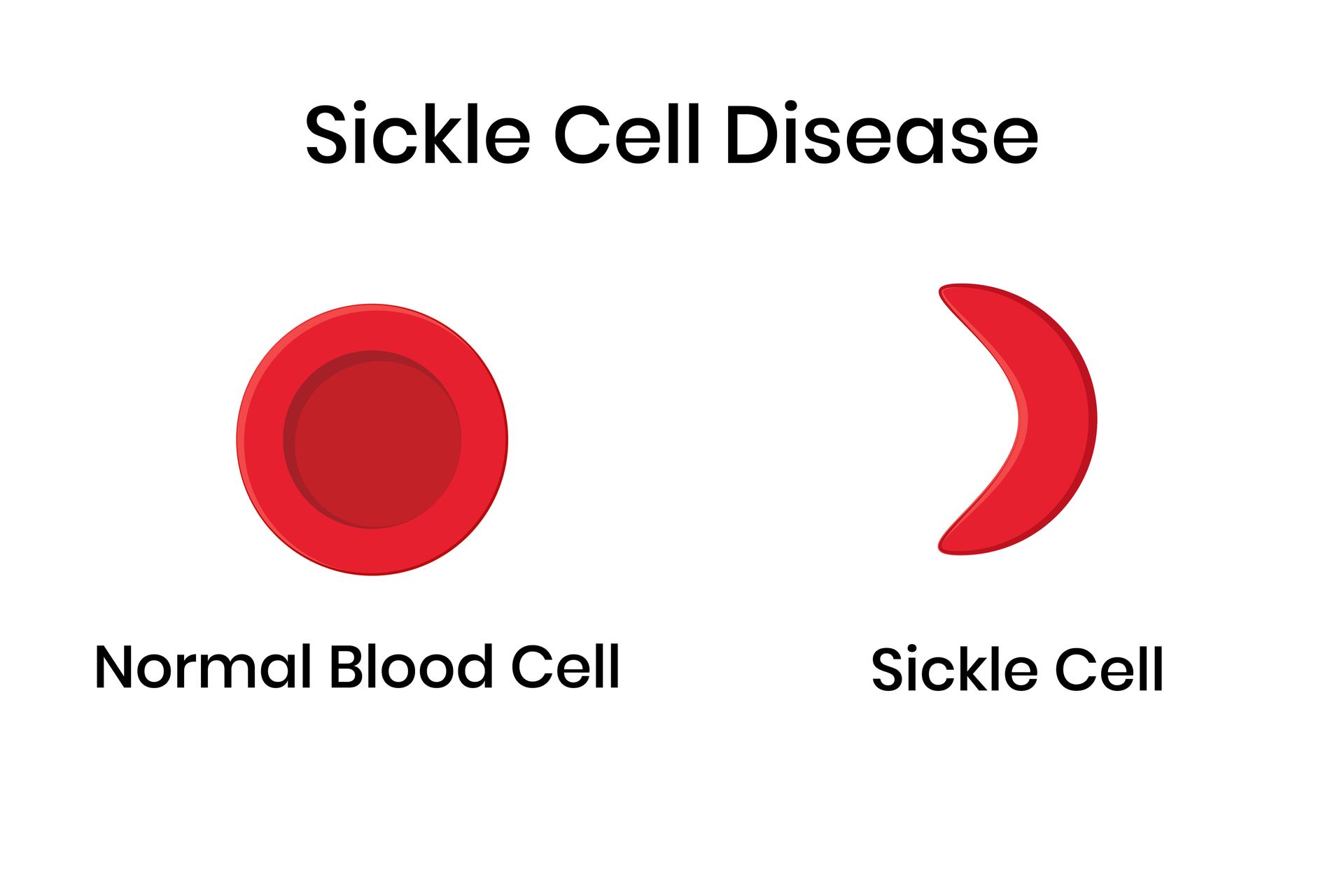
Sickle Cell Disease (SCD)
Phase 3 Study of Crizanlizumab
Who May Qualify:
- Diagnosed with sickle cell disease
- Experienced multiple pain crises
Purpose: Evaluating whether treatment may help reduce pain crises.
Acute Myeloid Leukemia (FLT3-ITD+)
Phase 2 Study of Quizartinib Maintenance Therapy
Who May Qualify:
- Diagnosed with AML (FLT3-ITD+)
- Currently in remission
- No prior stem cell transplant
- Candidate for maintenance therapy
Purpose: Evaluating safety and tolerability of maintenance therapy.
Check Your Eligibility
Complete this short form and our team will contact you.
Contact Information
3465 Nazareth Road, Suite 102
Easton, PA 18045
Phone: (833) 330-0930
Text: (610) 507-4795
Email: info@goodclinicaltrials.com
Disclaimer
Participation in a clinical trial is voluntary. Eligibility criteria apply. Study-related care is provided as part of the research protocol and may differ from standard medical care.